During the course of attending massage school at A New Beginning School of Massage, students are given a number of assignments that require research and writing. Some of these assignments result in very insightful and well thought out information and decision-making outcomes. I am happy to share some of their assignments for you to enjoy.
Preface:
Does massage therapy flush lactic acid from the muscles? Since I began working out and trading massage therapy I have heard that this is an important benefit of massage therapy. A good massage certainly feels wonderful after a strenuous work out. Who wouldn’t want that? Is there a mechanical action reaction from compressive and Effleurage, or Swedish touch that directly “flushes” the molecular compound called Lactic Acid “out” of each muscle cell? Where does this Lactic acid come from? Is it a result of a natural metabolic process as the cell produces energy during action? What happens to this Lactic acid and why? What could be the benefits of flushing or releasing lactic acid from the muscle? Where would it go? Is there a co relation to just feeling good, and a reduction of lactic acid in the muscle tissue? Is the pain associated from strenuous exercise a result of lactic acid in the muscle cells?
As professional massage therapists, it is incumbent upon us to inform the client of possible outcomes from massage therapy. This we do in a limited fashion in the assessment phase of the session. We ask the client what their expectations are from receiving massage therapy. We may be asked this question “Will this body work release lactic (and “toxins) from my cells?” To make an uninformed claim could lead to unintended consequences and a probably dissatisfied client whose expectations were not met, or are unobtainable. To build professional integrity we must advise the client with valid and substantive claims that meet their expectations. Claims that we know are true to the best of our knowledge. Our knowledge backed up by science and evidence. “We all have heard, or know that…..” is anecdotal and may lead the client to frustration and disappointment from the service. This is the last thing a professional Massage Therapist wants from their work.
Massage Therapy as an industry must have a system of ethics that adheres to information backed up by science just as the Medical industry is grounded in science. This derives from peer review papers based upon empirical scientific method, research, statistical evaluation as well as consensus of the industry. My intention with this paper will be to resolve some of the above questions in order to make an informed and valid statement regarding lactic acid and how massage therapy will help or not help the “build up” and release of this biological component of our metabolism.
I chose this topic because “I just do not know” the truth of this assertion. I would feel a lack of integrity in the informing of a client something that I could not validate. All things are connected and each part adds to or detracts from the building of the Massage Therapy industry. Our highest goal is to treat each client with the highest professional regard and respect for the best and greatest outcomes, every time a human being comes to us for healing or to just feel better. Knowledge is the basis of being a Professional, and this is my goal.
Thesis Statement
The current science indicates that massage does not actively “de tox” muscle tissue by “pushing out” from the muscle cells. I think that there is some effect as yet undefined but correlating to Well Being, feeling better as a result of massage therapy that may allow a homeostatic effect that enhances in the metabolic process of lactic genesis.
Outline
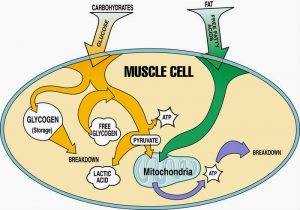
What is Lactic Acid?
Lactic acid is an organic compound with the formula CH3CH(OH)COOH. In its solid state, it is white and water-soluble. In its liquid state, it is clear. It is produced both naturally and synthetically. With a hydroxyl group adjacent to the carboxyl group, lactic acid is classified as an alpha-hydroxy acid (AHA). In the form of its conjugate base called lactate, it plays a role in several biochemical processes.
In solution, it can ionize a proton from the carboxyl group, producing the lactate ion CH
3CH(OH)CO−
2. Compared to acetic acid, its pKa is 1 unit less, meaning lactic acid deprotonates ten times more easily than acetic acid does. This higher acidity is the consequence of the intramolecular hydrogen bonding between the α-hydroxyl and the carboxylate group.
Lactic acid is chiral, consisting of two optical isomers. One is known as L-(+)-lactic acid or (S)-lactic acid and the other, its mirror image, is D-(−)-lactic acid or (R)-lactic acid. A mixture of the two in equal amounts is called DL-lactic acid, or racemic lactic acid.
Lactic acid is hygroscopic. DL-lactic acid is miscible with water and with ethanol above its melting point which is around 17 or 18 °C. D-lactic acid and L-lactic acid have a higher melting point.
In animals, L-lactate is constantly produced from pyruvate via the enzyme lactate dehydrogenase (LDH) in a process of fermentation during normal metabolism and exercise. It does not increase in concentration until the rate of lactate production exceeds the rate of lactate removal, which is governed by a number of factors, including monocarboxylate transporters, concentration and isoform of LDH, and oxidative capacity of tissues. The concentration of blood lactate is usually 1–2 mmol/L at rest, but can rise to over 20 mmol/L during intense exertion[4] and as high as 25 mmol/L afterward.[5]
In industry, lactic acid fermentation is performed by lactic acid bacteria, which convert simple carbohydrates such as glucose, sucrose, or galactose to lactic acid. These bacteria distilled water, generally in concentrations isotonic with human blood. It is most commonly used for fluid resuscitation after blood loss due to trauma, surgery, or burns. can also grow in the mouth; the acid they produce is responsible for the tooth decay known as caries.[6][7][8][9]
In medicine, lactate is one of the main components of lactated Ringer's solution and Hartmann's solution. These intravenous fluids consist of sodium and potassium cations along with lactate and chloride anions in solution with distilled water, generally in concentrations isotonic with human blood. It is most commonly used for fluid resuscitation after blood loss due to trauma, surgery, or burns. (Wikipedia)
Exercise and lactate
During power exercises such as sprinting, when the rate of demand for energy is high, glucose is broken down and oxidized to pyruvate, and lactate is then produced from the pyruvate faster than the body can process it, causing lactate concentrations to rise. The production of lactate is beneficial because it regenerates NAD+ (pyruvate is reduced to lactate while NADH is oxidized to NAD+), which is used up in oxidation of glyceraldehyde 3-phosphate during production of pyruvate from glucose, and this ensures that energy production is maintained and exercise can continue. (During intense exercise, the respiratory chain cannot keep up with the amount of hydrogen atoms that join to form NADH, and cannot regenerate NAD+ quickly enough.)
The resulting lactate can be used in two ways:
However, lactate is continually formed even at rest and during moderate exercise. Some causes of this are metabolism in red blood cells that lack mitochondria, and limitations resulting from the enzyme activity that occurs in muscle fibers having a high glycolytic capacity (Wikipedia)
Is the pain of strenuous exercise caused by Lactic Acid genesis?
In a Viewpoint article, Lindinger (2011) commented that ‘…lactate accumulation is a necessary evil associated with high speed’. Added commentary included, ‘That such high rates of lactate production can benefit performance, and yet such high lactate concentrations impair performance (Cairns, 2006), remains a conundrum of exercise physiology’. It is important to correct several immediate and underlying aspects of this interpretation of metabolic biochemistry and the data from the original research of Kitaoka et al. (2011).
Repeated intense muscle contraction can cause high rates of lactate production, with greatest rates occurring in fast-twitch glycolytic (type IIb) muscle as explained by Lindinger (2011). The long history of lactate, lactic acid and exercise-induced fatigue, even to the present time, has been tainted by erroneous interpretations of a detrimental view towards inter-relationships between muscle lactate production, acidosis and muscle contractile failure (Robergs et al. 2004, 2005; Cairns, 2006; Brooks, 2010). However, it is clear from application of fundamental principles of organic chemistry, enzyme kinetics and metabolic biochemistry that muscle lactate production is an essential feature of repeated intense muscle contractions, and that without lactate production such repeated contractions could not occur (Robergs et al.2004).
Why is lactate production essential? Muscle must produce lactate during intense contractions and/or when there are insufficient mitochondria and/or oxygen, because this remains the only alternative (non-mitochondrial) means to regenerate cytosolic NAD+. Muscle lactate production is therefore a rapid means to maintain cytosolic redox, which sustains phase II of glycolysis, which in turn sustains glycolytic ATP regeneration. Lactate production also metabolically consumes an H+ ion, which means that it directly opposes metabolic H+ release and cellular acidosis, which in turn means that muscle H+ release would be greater for a given rate of ATP turnover without lactate production (Robergs et al. 2004).
There is no ‘evil’ to muscle lactate production. As there is no clear evidence that lactate has any direct negative effect on muscle contraction, there is also no ‘conundrum’ to muscle lactate. Muscle lactate production is essential to sustained, repeated intense muscle contraction. Post-training increases in MCT1 and MCT4 support greater rates and total capacities of muscle lactate efflux during intense exercise performance, thereby allowing for a greater capacity for muscle lactate production and related glycolytic ATP turnover. It is long overdue, based on all aspects of the scientific method, to recognize muscle lactate production as a benefit to intense exercise performance and muscle biochemistry. I encourage researchers and educators alike to present an interpretation of muscle lactate production where the evidence-based view of the benefits of lactate production are espoused rather than a traditional blame of fatigue and acidosis simply because that is how it has always been. (Robergs et al. 2004)
Thus the aches and pain in strenuous exercise exist it is an indication of muscle fatigue but the formation of lactic acid facilitates extended muscle contraction and forestalls muscle failure.
Does massage therapy “flush” lactic acid from the muscle tissue?
True or false: massage after exercise assists in the removal of lactic acid. Answer: False.
The research overwhelmingly refutes this commonly held and frequently exclaimed belief. The lactic acid debate has raged for a century, and for the past two decades the research consistently demonstrates that blood lactate will return to normal within 20-60 minutes post-exercise, regardless of intervention. Yet, massage schools continue to teach this flawed lactic acid theory and massage therapists still declare it as truth to clients and the media.
It is time to reform our declaration on the benefits of massage for clients. It is essential to stop perpetuating a fasle theory. As a result of this pervasive myth, articles are published making damaging claims about massage, such as "research questions efficacy of massage as an aid to recovery in post exercise settings" and "massage not an effective treatment for enhancing long-term restoration of post-exercise muscle strength and its use in athletic settings should be questioned." Undocumented claims, such as "massage assists in the removal of lactic acid" draw attention and will be tested. The negative results become fodder for those looking for a shocking headline.
Even if massage does not help move lactic acid, thousands of elite athletes and trainers can't be wrong when they commit valuable resources to pack up massage therapists and take them across the country on bike races, or provide precious space in the medical tents at the Olympics, or allow the female massage therapist an unprecedented seat on the bench in the San Diego Padre's dugout. Massage does work. But, in an era of open access to research and the push for evidence-informed practices, we must heed the available data and speak accurately about what we do and why it works. When we fail to do so, it comes back and bites us where it hurts. We must better understand the known physiological effects of massage and how massage benefits our clients to be able to refute gross conclusions that don't accurately reflect our role in helping athletes recover from post-exercise symptoms.
In order to better understand why the belief that massage removes lactic is false, we need to better understand blood lactate and the causes of post-exercise symptoms. The first questions to explore are: what is lactic acid or blood lactate and what does it do? If lactic acid buildup doesn't cause muscle soreness and fatigue, then what does? Does massage enhance performance, relieve muscle fatigue, and reduce muscle soreness? If so, how? What do we know about the physiological effects of massage and what has yet to be demonstrated through research?
Once we have appraised the best available information, we can reform our statements about how massage benefits clients. Then we need to encourage all massage therapists to promote the best available data on the benefits of massage to clients. Finally, it is important to inform the next generation of research to encourage questions meaningful to practitioners. We can submit questions that we confront regularly in practice, identify the theories we dispute, and hopefully provoke someone to explore potential answers. (Massage Bodywork, Diana Thompson 2011).
Conclusion
The genesis of lactic acid, lactate, in muscle cells and tissue is an integral component of muscle contraction that facilitates continued muscle use even in anaerobic muscle fatigue. The metabolic process produces lactate as a function of anaerobic combustion of glucose to continue the generation of ATP that in turn provides energy to muscle tissue for repeated contractions. This continues until muscle fatigue leads to muscle failure until the aerobic process resumes balance in the cells, that is to say in recovery.
The application of Swedish or deep pressure or contractions, massage on muscle groups or single muscle will allow the muscle to relax and recover in an enhanced state, but still in the presence of lactate. There is no scientific evidence that shows or proves that massage alone will “push out” this by-product of cellular combustion to speed muscle recovery or that compression, Swedish or deep massage will reduce lactic acid at the cellular level that leads to pain or stiffness relief. Yes, massage therapy will help the body feel better, but does not mean it mechanically reduces lactic acid content.
References:
Robergs, R. A. (2011), Nothing ‘evil’ and no ‘conundrum’ about muscle lactate production. Experimental Physiology, 96: 1097–1098. doi:10.1113/expphysiol.2011.057794
Thompson, D. L. (2011). The Lactic Acid Debate. Massage and Bodywork Magazine. Retrieved from https://www.abmp.com/textonlymags/article.php?article=173.